
Copovidonum25086-89-9
Product Introduction
Copovidonum25086-89-9 (CAS 25086-89-9) is a vinylpyrrolidone-vinyl acetate copolymer (60:40 ratio) meeting Ph.Eur/USP-NF standards. This pharmacopeial polymer (K-value 25-35) functions as solubility enhancer and tablet binder in solid dosage formulations.
Povidone VA64 is generally considered safe for use in pharmaceutical and cosmetic applications, with no known toxicological effects. In fact, it is often used as a replacement for other excipients, such as polyethylene glycol, which have been shown to have potential toxic effects. It is also biocompatible, meaning that it does not cause a significant immune response in the body.
Overall, Povidone VA64 is a versatile excipient that has many applications in various industries. Its unique properties make it an ideal choice for tablet formulations, coatings, and a range of other applications. As technology and research continue to improve, we can expect to see even more applications for Povidone VA64 in the future.
Critical Pharmaceutical Applications
Instant-release tablets
Disintegration accelerator (<5 min dissolution)
Solid dispersions
Bioavailability booster for BCS Class II/IV APIs
Melt extrusion
Matrix former for hot-melt extrusion (HME) processing
Film coatings
Moisture barrier for hygroscopic actives
Direct compression
Dry binder improving tablet friability (<0.8%)
Release Specifications
|
Parameter |
Specification |
|
CAS No |
25086-89-9 |
|
VP:VA Ratio |
60:40 ± 2% (NMR verified) |
|
K-value |
28-32 (1% in ethanol) |
|
Residual Monomers |
≤0.1% NVP / ≤0.1% VAc (GC) |
|
Loss on Drying |
≤5.0% (105°C, 3h) |
|
Compliance |
Ph.Eur 10.0 / USP-NF <1057> |
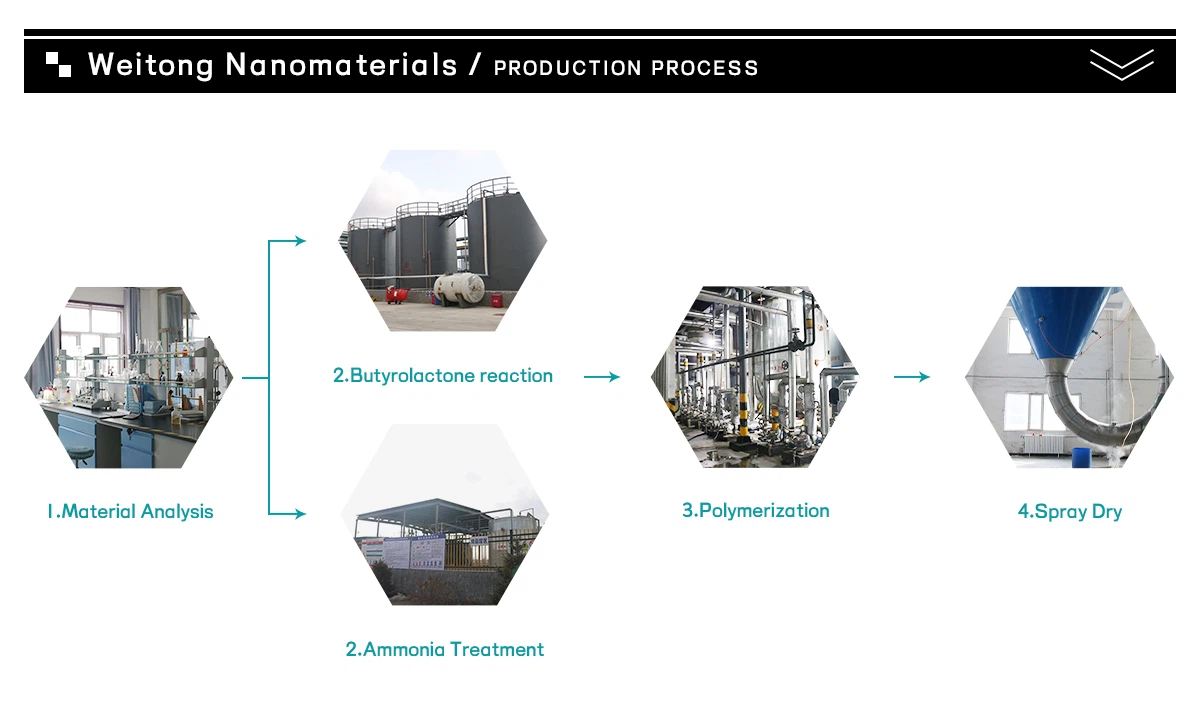
Processing Protocol
Regulatory support
CEP available for EU markets
01
Batch traceability
Full residual solvents report (ICH Q3C)
02
Scale flexibility
R&D samples to 15 MT production batches
03
Technical data
Includes DSC thermograms for HME parameters
04
Advantages to choose us:
1.Free sample service: We can provide you with free samples if you want.
2.Direct factory,competitive price: We are the manufacturer, and we produce these products by ourselves, so there is no extra payment link in the middle. We can give you the most competitive price.
3.Quality control: We have the ISO9001 certificate, and we strictly follow GMP production standards for production.
4.Good after-sale service: We have a sound after-sales service system, so whatever you have any doubt about the product ,you can strictly contact us , we will give you a satisfied plan.
5.Regulatory support: CEP available for EU markets
6.Batch traceability: Full residual solvents report (ICH Q3C)
7.Scale flexibility: R&D samples to 15 MT production batches
8.Technical data: Includes DSC thermograms for HME parameters
9. What can you get from us?
You can get excellent products (strict quality control ) ,factory direct sale (favorable and competitive price),great service( after-sales services, fast delivery)and professional business consultation.



Hot Tags: Copovidonum25086-89-9, China Copovidonum25086-89-9 manufacturers, suppliers, factory, Polyvinylpyrrolidone Pvp K30 Solution, Copovidone, Copovidonum, 1 4 Butanediol Liquid, N-methylpyrrolidone
Previous
Va 64 25086-89-9You Might Also Like
Send Inquiry









